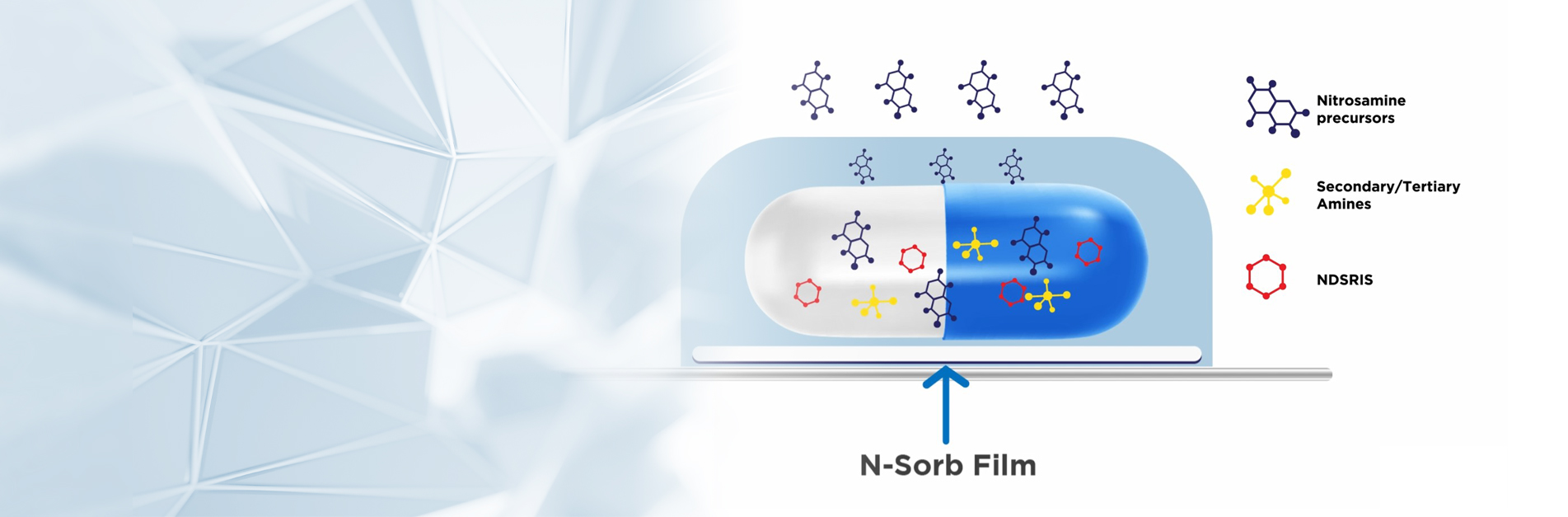
Aptar Active Material Science is the premier active material science solutions expert that transforms ideas into market opportunities, accelerates and de-risks the product development process, and provides complete solutions that improve consumers’ and patients’ lives.
Based on breakthrough science, Aptar’s proprietary 3-Phase Activ-Polymer™ technology enables the development of innovative, highly engineered material science based solutions to meet a broad range of product stability needs across six key therapeutic areas. We offer a complete spectrum of capabilities to support our customers and expedite speed-to-market.